Mon - Sat: 9:30 - 18:00
Blood Bag Testing – Ensuring Safety,
Sterility & Performance for Life-Saving
Applications
Blood bag testing is a critical quality assurance process that ensures the safety, sterility, and functional integrity of blood collection, storage, and transfusion systems. Given the direct impact on patient health, every component—from the bag material to the tubing and ports—must comply with ISO 3826, BIS, and other global regulatory standards. At SIMA LABS, we conduct comprehensive testing that covers mechanical strength, chemical compatibility, sterility validation, pyrogen testing, and leachable analysis. Our advanced testing ensures that blood bags maintain the viability of stored blood, prevent contamination, and perform reliably during clinical use.
Why Choose SIMA LABS for Blood Bag Testing?
- Global Standards Compliance – ISO 3826, BIS, WHO, and FDA guidelines.
- Comprehensive Evaluation – Physical, chemical, microbiological, and functional performance testing.
- Advanced Testing Facilities – Equipped for sterility, pyrogen, and leachables analysis.
- Material Safety Checks – Ensures non-toxicity and chemical compatibility with stored blood.
- Regulatory Support – Detailed reports for product registration, CE marking, and export compliance.
- Proven Expertise – Years of experience in medical device and transfusion product validation.
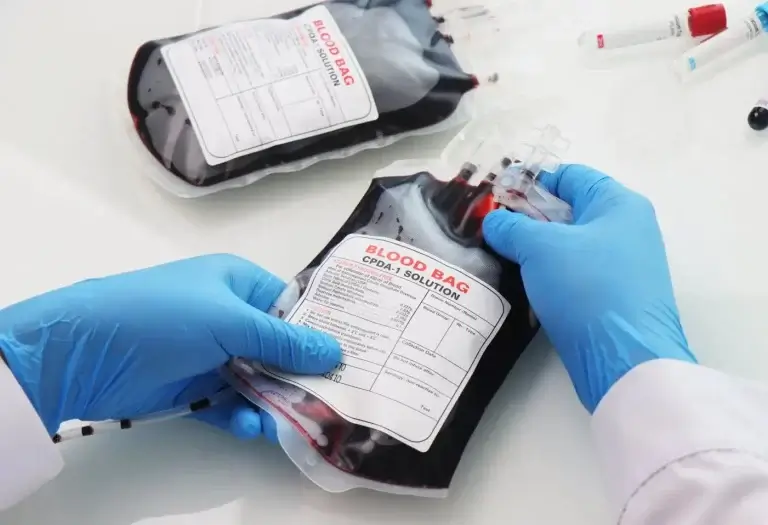
35+ Years of SIMA LABS – Blood Bag Testing You Can Trust
SIMA LABS is NABL accredited and approved under BIS for comprehensive testing of blood bags in compliance with ISO 3826, BIS standards, WHO guidelines, and FDA requirements. Our testing ensures that blood collection, storage, and transfusion systems meet strict safety, sterility, and performance benchmarks for both domestic and international healthcare markets.
We provide end-to-end testing for blood bags to ensure their quality, safety, and functionality in critical medical applications.
Scope of Testing:
Physical & Mechanical Testing:
Tensile strength of tubing and seals
Puncture and leak resistance
Drop impact testing
Port and closure integrity
Chemical Testing:
Extractables & leachables analysis
Plasticizer (DEHP) content testing
pH and chemical compatibility with blood
Microbiological Testing:
Sterility testing (ISO 11737)
Pyrogen and endotoxin testing (LAL method)
Functional Testing:
Volume accuracy and flow rate
Anticoagulant performance evaluation
Regulatory Compliance:
ISO 3826:2019 – Plastics containers for human blood and blood components
BIS specifications for blood bags
WHO prequalification requirements
FDA and CE Marking standards
With state-of-the-art testing infrastructure, validated protocols, and expert medical device analysts, SIMA LABS ensures your blood bags are safe, compliant, and ready for clinical use worldwide.
Trusted by leading brands
our customers are the driving force behind our growth and innovation







































Precision. Compliance. Innovation
Our Expertise
At Sima Labs, we specialize in advanced analytical testing, regulatory compliance, and quality assurance across the food, pharmaceutical, and environmental sectors—delivering reliable results with international standards and technical excellence.

Residue Analysis
SIMA LABS offers residue testing and compliance with ELV, RoHS, REACH, and EN71 standards

Microbiological Analysis
SIMA LABS provides microbiological testing and indoor air quality monitoring.

Molecular Biological Analysis
SIMA LABS offers GMO, allergen, pathogen testing, event identification, and pharma analysis for safety and compliance.

Method Development and Validation Studies
SIMA LABS provides stability testing, drug analysis, quality checks, lab consultancy, training, and R&D support.

Environment Monitoring
SIMA LABS offers environmental testing for pollutants, gases, and air quality monitoring.


